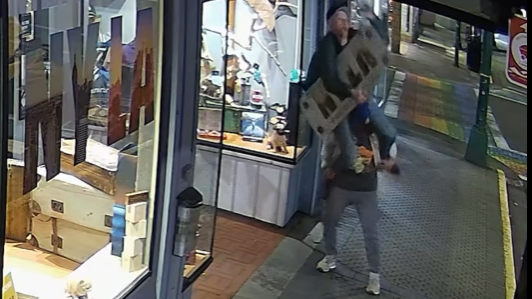Tottenham mom hopeful with new drug approval for cystic fibrosis
It's been a long road for Sasha Haughian, whose two sons live with cystic fibrosis (CF).
"I just never thought we would get this far," the Tottenham mom says after receiving news that Health Canada gave the green light to a life-saving new drug to treat CF, called Trikafta.
Both her boys, Andre, 11, and Joshua, 9, were diagnosed with CF, a genetic disease that has no cure and affects the digestive system and lungs.
CF is a progressive disease that can take a toll. Andre and Joshua live with a chronic cough that can keep them up nights.
Andre suffered a lung collapse and has permanent lung damage.
Haughian says she's hopeful with the prospect of a new drug.
"The bad days they're in the hospital, and I hope with these new drugs, those days are going to be few and far between."
For roughly two years, Haughian and families like hers have been fighting to get the drug and others like it approved.
Related Article: Mother fights for medication for her sons diagnosed with fatal disease
Cystic Fibrosis Canada chief scientific officer Dr. John Wallenburg says getting a new drug approved is no easy task. "Sadly, it's a rare occasion when a drug moves this smoothly and quickly through the system, and it really is a credit, I think, to the cystic fibrosis community."
People 12 and older will become eligible to receive Trikafta. Two other medications are also in the process of becoming widely accessible, but there are still a couple of hurdles left to go.
"The premiers and the health ministers across the country, we really need them to go 'Yes, this drug is a priority' and to list it on their provincial formularies as soon as possible," Dr. Wallenburg adds.
"It's like a big weight lifted off our shoulders, probably similar to when people got the vaccine or knew the vaccine was coming because you can just start to plan your future," Haughian concludes.
CTVNews.ca Top Stories

Quebec nurse had to clean up after husband's death in Montreal hospital
On a night she should have been mourning, a nurse from Quebec's Laurentians region says she was forced to clean up her husband after he died at a hospital in Montreal.
Northern Ont. lawyer who abandoned clients in child protection cases disbarred
A North Bay, Ont., lawyer who abandoned 15 clients – many of them child protection cases – has lost his licence to practise law.
Bank of Canada officials split on when to start cutting interest rates
Members of the Bank of Canada's governing council were split on how long the central bank should wait before it starts cutting interest rates when they met earlier this month.
Maple Leafs fall to Bruins in Game 3, trail series 2-1
Brad Marchand scored twice, including the winner in the third period, and added an assist as the Boston Bruins downed the Toronto Maple Leafs 4-2 to take a 2-1 lead in their first-round playoff series Wednesday
Cuban government apologizes to Montreal-area family after delivering wrong body
Cuba's foreign affairs minister has apologized to a Montreal-area family after they were sent the wrong body following the death of a loved one.
'It was instant karma': Viral video captures failed theft attempt in Nanaimo, B.C.
Mounties in Nanaimo, B.C., say two late-night revellers are lucky their allegedly drunken antics weren't reported to police after security cameras captured the men trying to steal a heavy sign from a downtown business.
What is changing about Canada's capital gains tax and how does it impact me?
The federal government's proposed change to capital gains taxation is expected to increase taxes on investments and mainly affect wealthy Canadians and businesses. Here's what you need to know about the move.
New Indigenous loan guarantee program a 'really big deal,' Freeland says at Toronto conference
Canada's Deputy Prime Minister Chrystia Freeland was among the 1,700 delegates attending the two-day First Nations Major Projects Coalition (FNMPC) conference that concluded Tuesday in Toronto.
'Life was not fair to him': Daughter of N.B. man exonerated of murder remembers him as a kind soul
The daughter of a New Brunswick man recently exonerated from murder, is remembering her father as somebody who, despite a wrongful conviction, never became bitter or angry.